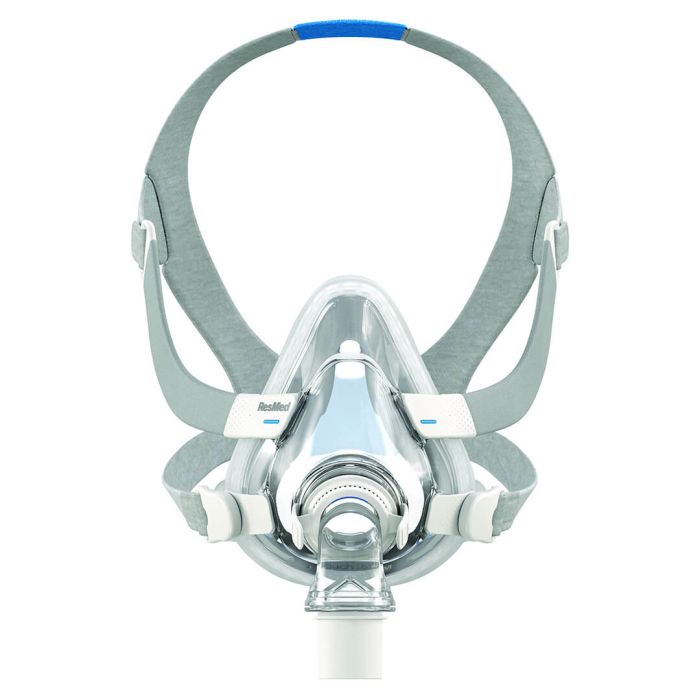
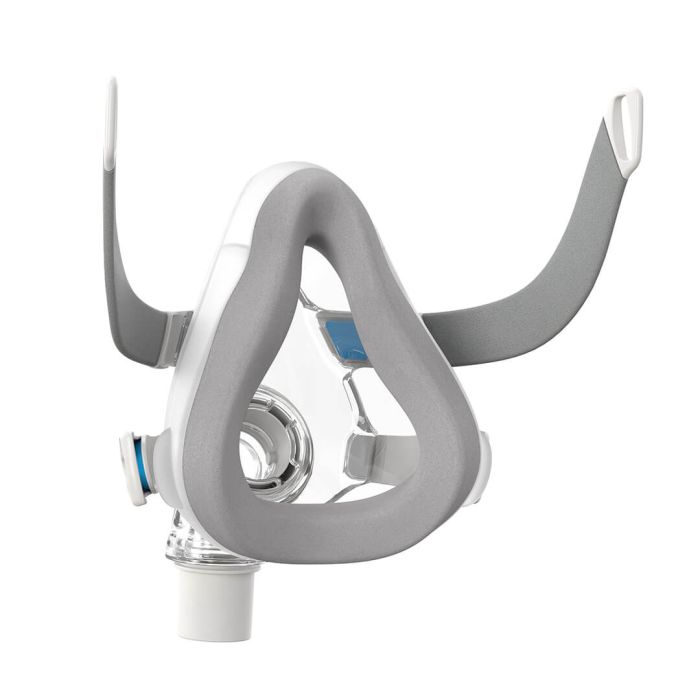
AirTouch F20 CPAP full face mask
The AirTouch F20 Face Mask features a comfortable UltraSoft memory foam cushion. Whisper-quiet to help you sleep, you can count on a reliable seal for a wide range of treatment pressures. Manufacturer recommandation is to replace the cushion after 1 to 2 months of use or at the first signs of deterioration.
SAFETY NOTICE: Prior to purchasing this mask, please verify that you do not have any of the contraindications on masks with magnets.
Professional advice - Full face CPAP mask
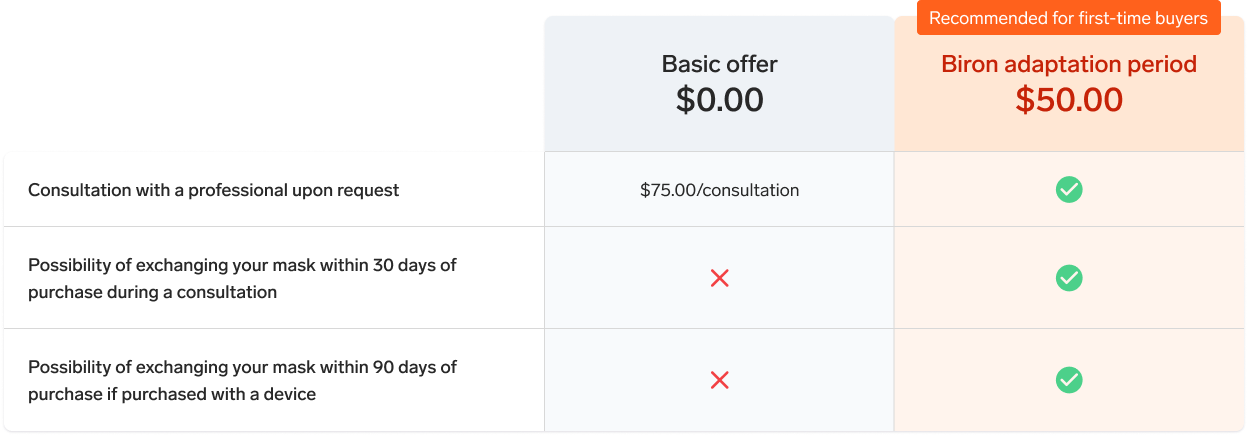
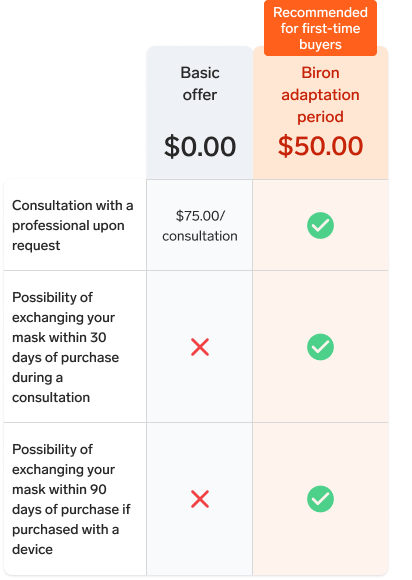
This full face mask is particularly recommended for people who breathe through their mouth or have certain medical conditions. If you are new to this type of mask, it is important to know that full face masks can, in some cases, affect the comfort or effectiveness of treatment. Feel free to chat with a sleep navigator to help you choose the model best suited to your situation.
This product is only refundable if it remains unopened. For more information, please read our Terms and conditions for online sales.
Features
Minimalist
Sensitive skin
Stability
Your device has been successfully added to the cart
Your mask has been successfully added to your cart
Exclusive, hassle-free deals for your device
The mask is not included with the device and is required for use.
Do you want to browse the masks?
Availability: Shipping within 3-5 business days